Efficacy of SAMSCA® (tolvaptan): Data from the pivotal clinical trials
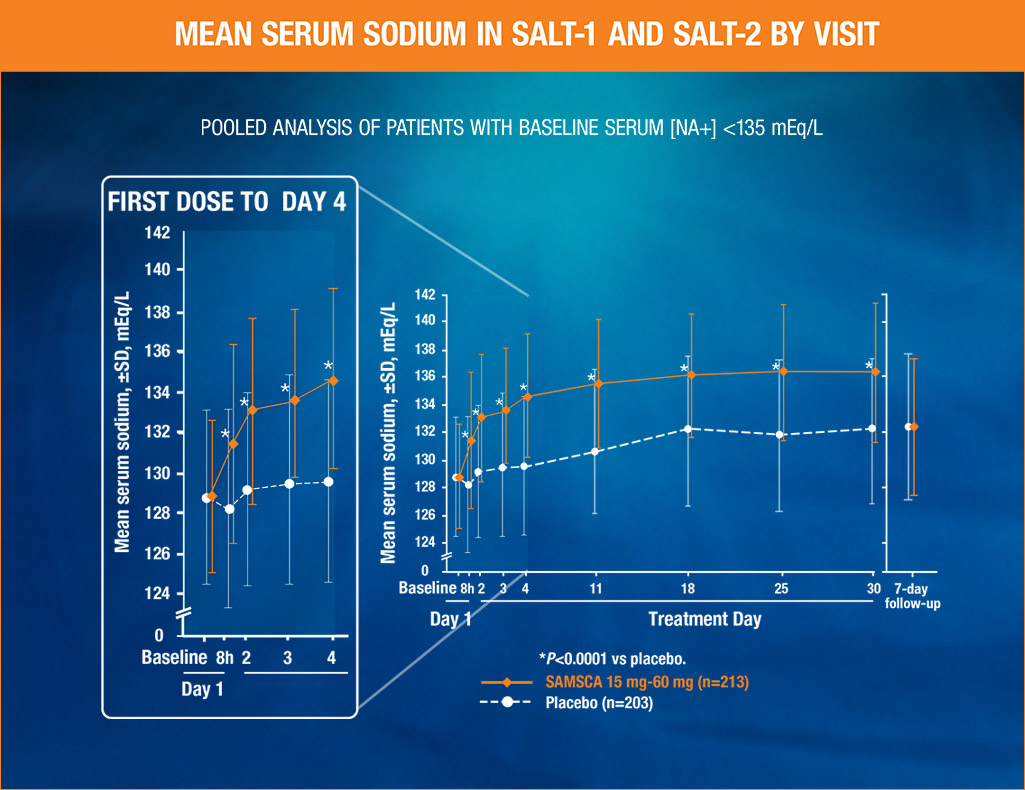
SAMSCA significantly increased average daily serum sodium from baseline to Day 4 and baseline to Day 30, vs placebo (P<0.0001) in the SALT (Study of Ascending Levels of Tolvaptan in hyponatremia) trials
- Primary end point was average daily AUC for change in serum sodium from baseline to Day 4 (tolvaptan [n=213], 4.0 mEq/L; placebo [n=203], 0.4 mEq/L [P<0.0001]) and baseline to Day 30 (tolvaptan [n=213], 6.2 mEq/L; placebo [n=203], 1.8 mEq/L [P<0.0001])
In the SALT (Study of Ascending Levels of Tolvaptan in hyponatremia) pivotal trials—two identical, 30-day, randomized, double-blind, placebo-controlled, multicenter studies—424 patients with euvolemic or hypervolemic hyponatremia (serum sodium <135 mEq/L) were treated for 30 days with tolvaptan or oral placebo, then followed for an additional 7 days after withdrawal. Primary end point from pivotal clinical trials was average daily AUC for change in serum sodium from baseline to Day 4 (tolvaptan, 4.0 mEq/L; placebo, 0.4 mEq/L, P<0.0001) and baseline to Day 30 (tolvaptan, 6.2 mEq/L; placebo, 1.8 mEq/L, P<0.0001). Mean change in serum sodium from baseline to 8 hours for tolvaptan, 2.5 mEq/L; placebo, -0.5 mEq/L, P<0.0001 (secondary end point). Starting dose was 15 mg, and was increased to 30 or 60 mg, if necessary. Within 7 days of discontinuation, serum sodium concentrations in tolvaptan-treated patients declined to placebo-like levels.
SAMSCA provided a statistically significant increase from baseline (P<0.0001) in serum sodium in patients with euvolemic or hypervolemic hyponatremia compared with placebo
- Significant increase in as early as 8 hours (mean change from baseline, secondary endpoint)
- Proven in two identical randomized, placebo-controlled, double-blind phase 3 studies (Study of Ascending Levels of Tolvaptan in hyponatremia 1 and 2) [SALT-1 and SALT-2]1
- The mean increases in serum sodium during treatment initiation (first 24 hours) were 4.06 mEq/L for SAMSCA 15 mg and 0.33 mEq/L for placebo (baseline <135 mEq/L)2
Too-rapid correction of serum sodium (e.g., >12 mEq/L/24 hours) can cause serious neurologic sequelae, including osmotic demyelination syndrome (ODS)
Post hoc analysis
- Post hoc analysis was done in 2 subgroup populations:
- Hyponatremia in SIADH subgroup population n=1103
- Hyponatremia in SIADH in patients with malignancy subgroup population n=284
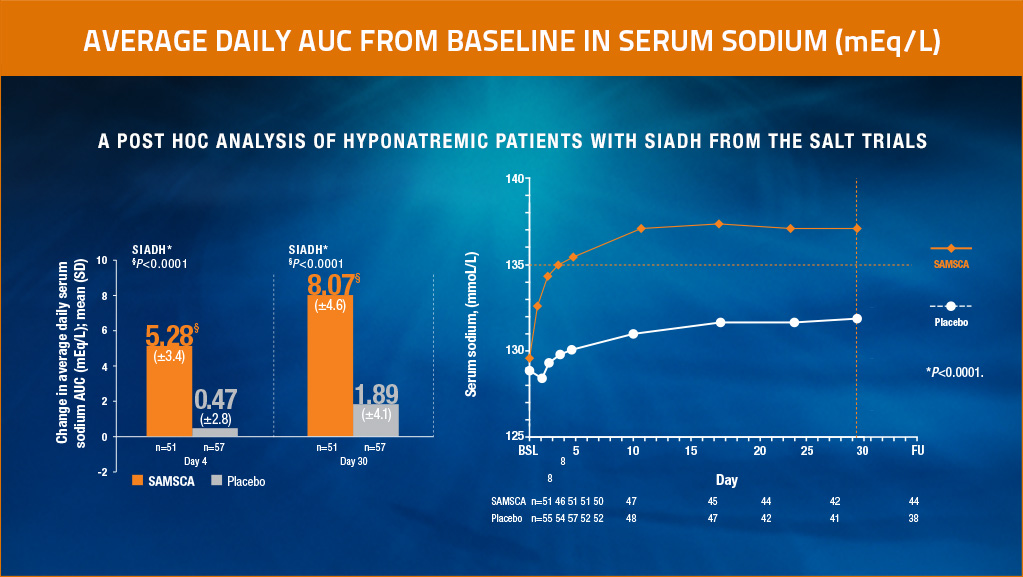
A post hoc analysis of hyponatremic patients with SIADH from the SALT trials
Significant increase in average daily serum sodium from baseline to Day 4 and to Day 30, and in as early as 8 hours for SAMSCA vs placebo3,4
Average daily AUC of change from baseline in serum sodium (last observation carried forward, left) and serum sodium (patients on treatment, right) in the combined hyponatremia in SIADH subgroups of SALT-1 and SALT-2. Results are consistent with those of the overall population in SALT-1 and SALT-2.
Adapted from Verbalis et al, Eur J Endocrinol, 2011.3
Average daily AUC of change from baseline in serum sodium (last observation carried forward, left) and serum sodium (patients on treatment, right) in the combined hyponatremia in SIADH subgroups of SALT-1 and SALT-2. Results are consistent with those of the overall population in SALT-1 and SALT-2.
Results from a post hoc analysis of 110 patients (n=52, SAMSCA; n=58, placebo) from the SALT-1 and SALT-2 trials with investigator-diagnosed SIADH3
- Primary end point: Significant change in average daily AUC for serum sodium from baseline to Day 4 (SAMSCA 5.28 mEq/L vs placebo 0.47 mEq/L; P<0.0001) and from baseline to Day 30 (SAMSCA 8.07 mEq/L vs placebo 1.89 mEq/L; P<0.0001)3
- Secondary end point: Significant change in mean serum sodium concentration from baseline to 8 hours (SAMSCA 4.0 mEq/L vs placebo -0.7 mEq/L; P<0.0001; n=102)4
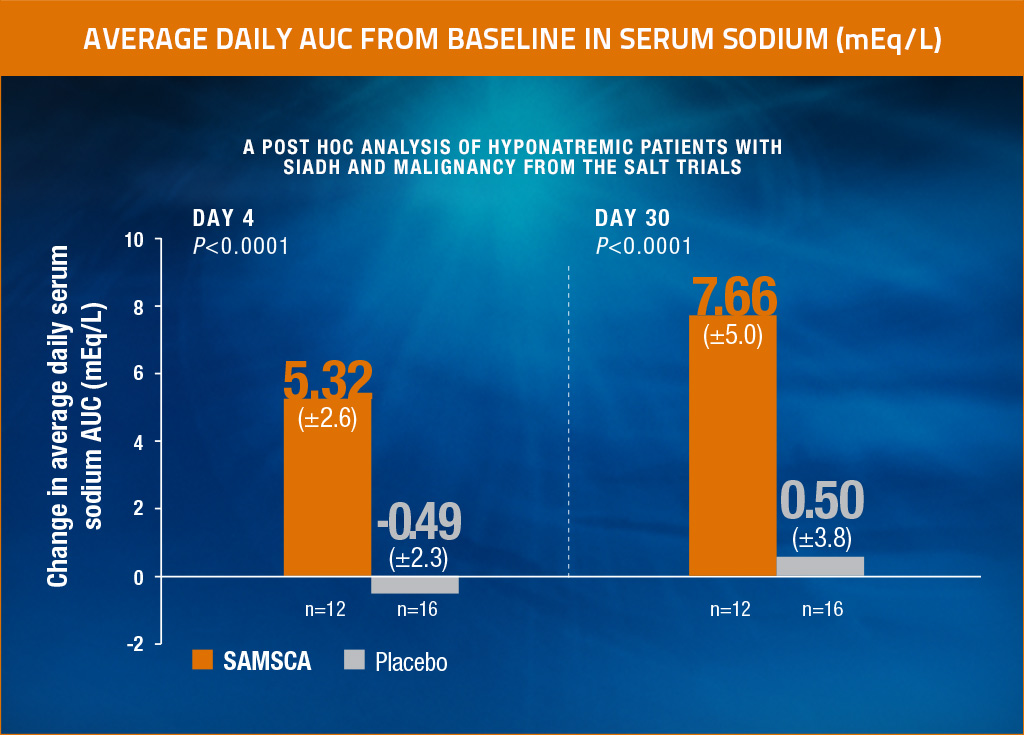
A post hoc analysis of hyponatremic patients with SIADH and malignancy from the SALT trials
Significant increase in average daily serum sodium from baseline to Day 4 and to Day 30, and in as early as 8 hours, for SAMSCA vs placebo4
This separate post hoc analysis in 28 hyponatremic patients with SIADH and malignancy (n=12, SAMSCA; n=16, placebo) from the SALT trials was conducted to determine the efficacy of SAMSCA in this group relative to the overall hyponatremia in SIADH population.
Last observation carried forward data shown. Similar results were shown for the subgroup of hyponatremic patients with SIADH and for the full population of patients included in SALT-1 and SALT-2. This separate post hoc analysis in 28 hyponatremic patients with SIADH and malignancy (n=12, SAMSCA; n=16, placebo) from the SALT trials was conducted to determine the efficacy of SAMSCA in this group relative to the overall hyponatremia in SIADH population.
Average daily AUC of change from baseline in serum sodium in hyponatremic patients with SIADH and malignancy from SALT-1 and SALT-2. Results are consistent with those of the overall population in SALT-1 and SALT-2.
The post hoc analysis of patients in the trial was not powered to analyze the hyponatremic patients with SIADH and malignancy separately.
Results from a post hoc analysis of 28 patients (n=12, SAMSCA; n=16, placebo) from the SALT-1 and SALT-2 trials with investigator-diagnosed SIADH and malignancy4
- Primary end point: Significant change in average daily AUC for serum sodium from baseline to Day 4 (SAMSCA 5.32 mEq/L vs placebo -0.49 mEq/L; P<0.0001) and from baseline to Day 30 (SAMSCA 7.66 mEq/L vs placebo 0.50 mEq/L; P<0.0001).4
- Secondary end point: Significant change in mean serum sodium from baseline to 8 hours (SAMSCA 4.6 mEq/L vs placebo -1.6 mEq/L; P<0.0001; n=25).4
In a separate small clinical trial that studied SAMSCA in hospitalized cancer patients with nonhypovolemic hyponatremia, efficacy and safety results were consistent with SALT-1 and SALT-2 results5
- This prospective, randomized, placebo-controlled, double-blind trial studied SAMSCA use in adult patients with cancer who were admitted to The University of Texas MD Anderson Cancer Center with nonhypovolemic hyponatremia (125-130 mEq/L; N=48).
- Subjects were randomized to receive either SAMSCA or placebo. Both groups received the standard of care for hyponatremia, except that patients were allowed to drink in response to thirst
- 94% of patients (16/17) treated with SAMSCA achieved the primary end point of serum sodium correction (≥136 mEq/L) on day 14, vs 8% (1/13) on placebo (P<0.001)
- Efficacy and safety results were consistent with SALT-1 and SALT-2 results

The SAMSCA MOA video illustrates the relationship between vasopressin and hyponatremia, as well as the potential benefits of vasopressin V2-receptor antagonism.