Efficacy of SAMSCA® (tolvaptan): Data from the pivotal clinical trials
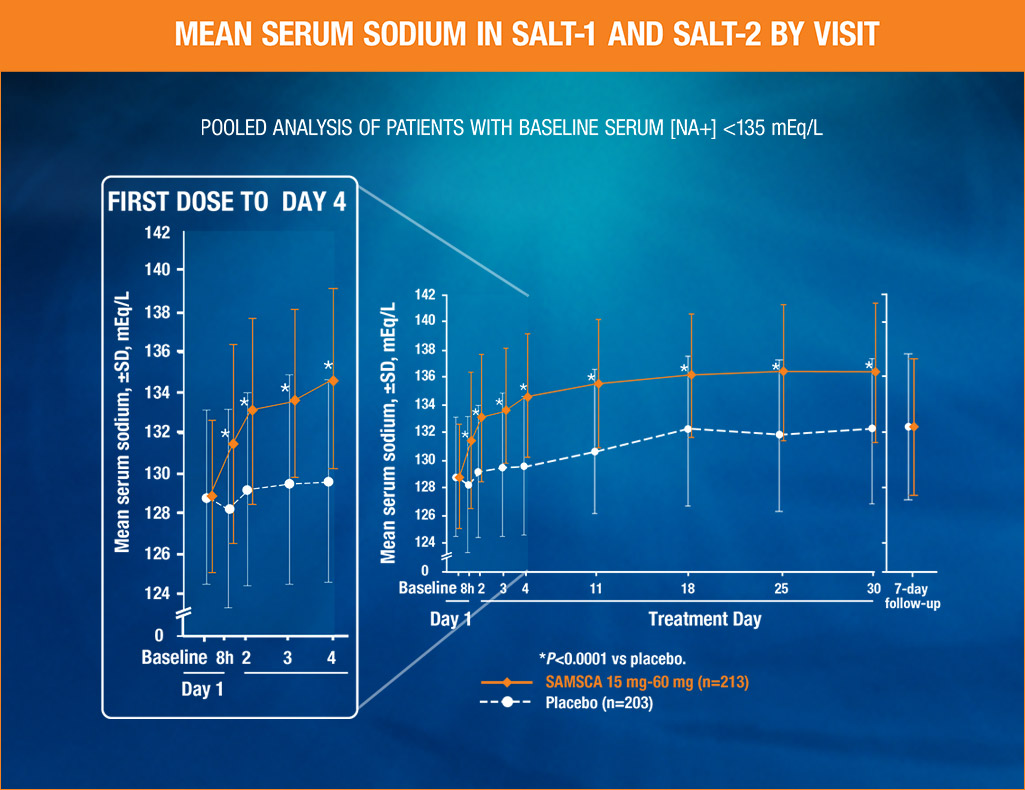
SAMSCA significantly increased average daily serum sodium from baseline to Day 4 and baseline to Day 30, vs placebo (P<0.0001) in the SALT (Study of Ascending Levels of Tolvaptan in hyponatremia) trials
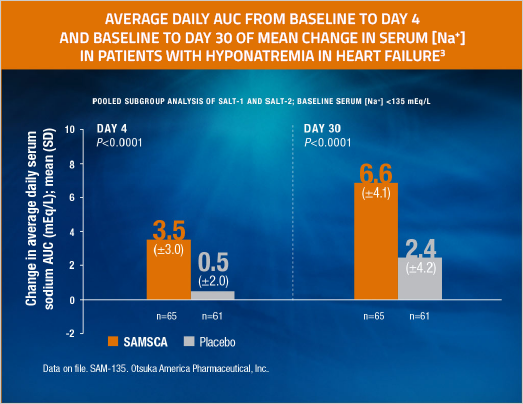
- Primary end point was average daily AUC for change in serum sodium from baseline to Day 4 (tolvaptan [n=213], 4.0 mEq/L; placebo [n=203], 0.4 mEq/L [P<0.0001]) and baseline to Day 30 (tolvaptan [n=213], 6.2 mEq/L; placebo [n=203], 1.8 mEq/L [P<0.0001])
In the SALT (Study of Ascending Levels of Tolvaptan in hyponatremia) pivotal trials—two identical, 30-day, randomized, double-blind, placebo-controlled, multicenter studies—424 patients with euvolemic or hypervolemic hyponatremia (serum sodium <135 mEq/L) were treated for 30 days with tolvaptan or oral placebo, then followed for an additional 7 days after withdrawal. Primary end point from pivotal clinical trials was average daily AUC for change in serum sodium from baseline to Day 4 (tolvaptan, 4.0 mEq/L; placebo, 0.4 mEq/L, P<0.0001) and baseline to Day 30 (tolvaptan, 6.2 mEq/L; placebo, 1.8 mEq/L, P<0.0001). Mean change in serum sodium from baseline to 8 hours for tolvaptan, 2.5 mEq/L; placebo, -0.5 mEq/L. P<0.0001 (secondary end point). Starting dose was 15 mg, and was increased to 30 or 60 mg, if necessary. Within 7 days of discontinuation, serum sodium concentrations in tolvaptan-treated patients declined to placebo-like levels.
Compared with placebo, SAMSCA provided a statistically significant increase from baseline (P<0.0001) in serum sodium in patients with euvolemic or hypervolemic hyponatremia
- Significant increase in as early as 8 hours (mean change from baseline, secondary endpoint)
- Proven in two identical randomized, placebo-controlled, double-blind phase 3 studies (Study of Ascending Levels of Tolvaptan in hyponatremia 1 and 2)1
- The mean increases in serum sodium during treatment initiation (first 24 hours) were 4.06 mEq/L for SAMSCA 15 mg and 0.33 mEq/L for placebo (baseline <135 mEq/L)2
Too-rapid correction of serum sodium (e.g., >12 mEq/L/24 hours) can cause serious neurologic sequelae, including osmotic demyelination syndrome (ODS).
SAMSCA significantly increased serum sodium in hyponatremic patients with heart failure
- Demonstrated in the pooled subgroup analysis of SALT-1 and SALT-2

The SAMSCA MOA video illustrates the relationship between vasopressin and hyponatremia, as well as the potential benefits of vasopressin V2-receptor antagonism.